The Health Technical Memorandum (HTM) 01-01 explains the management of Decontamination, and the various ways to sterilise reusable medical devices used in acute care.
The Health Technical Memorandum aims to improve the safety of patients, the clinical effectiveness and patient experience, by encouraging healthcare providers and suppliers to adhere to best practice guidelines set by the Department of Health.
What is the HTM?
The HTMs give comprehensive advice and guidance on the design, installation and operation of specialised building and engineering technology used in the delivery of Healthcare.
The focus of the HTM guidance remains on Healthcare-specific elements of standards, policies and up-to-date established best practice.
The providers of Healthcare services have a duty of care to ensure that appropriate governance arrangements are in place, and that they are managed effectively. The Health Technical Memorandum series provides best practice engineering standards and policy to enable management of this duty of care.
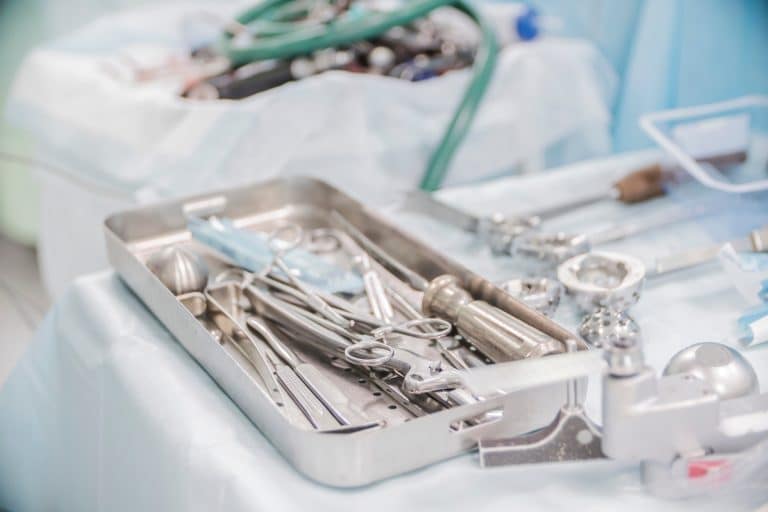
What is the HTM-01-01?
The HTM-01-01 explains the management of Decontamination, and the various ways to sterile reusable medical devices used in acute care.
Part A covers the policy, management approach and choices available in formulating a locally developed, risk-controlled operational environment. The technical concepts are based on European (EN), International (ISO) and British (BS) Standards used alongside policy and broad guidance. In addition to the prevention of transmission of conventional pathogens, precautionary procedures in respect of human prion diseases, including variant Creutzfeldt-Jakob disease (vCJD), are clearly stated. Advice is also given on surgical instrument management related to surgical care efficiencies and contingency against perioperative non-availability of instruments.
Part B covers common elements that apply to all methods of surgical instrument reprocessing, such as:
• Test equipment and materials
• Design and pre-purchase considerations
• Validation and verification.
Part C covers standards and guidance on steam sterilisation.
Part D covers standards and guidance on washer-disinfectors.
Part E covers low temperature (non-steam) sterilisation processes (such as the use of vapourised hydrogen peroxide gas plasmas and ethylene oxide exposure).

Why is the HTM-01-01 so important in Decontamination?
The HTM-01-01 is the regulation followed by the Healthcare industry. It outlines all the legal requirements that Sterile Services must comply with in order to remain operational.
What does Sychem offer?
At Sychem, we offer a complete portfolio of Decontamination equipment, Infection Control consumables and Servicing options, to support SSD departments in their continued compliance with the HTM-01-01.
Capital equipment
Our portfolio includes various Decontamination capital equipment, manufactured by our partner Steelco. Our vast range comprises steam sterilising autoclaves, low-temperature sterilisers, washer disinfectors and ultrasonic washers.
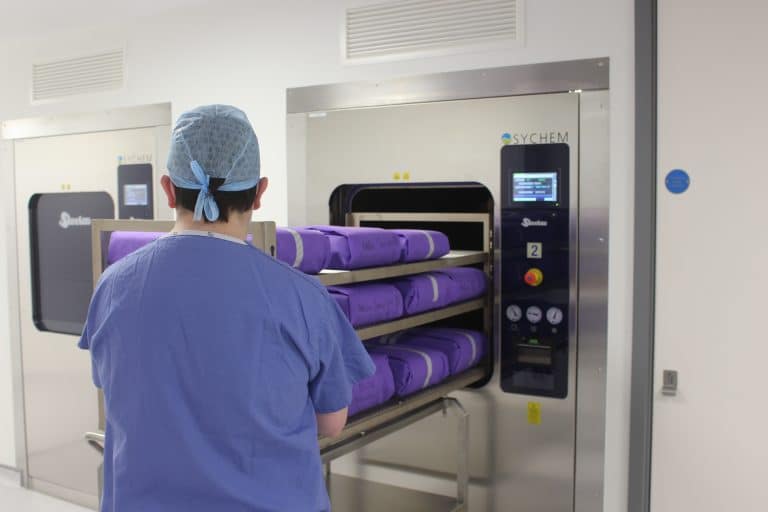
Infection Control consumables
We also offer several Infection Control consumables, or Process Challenge Devices, by our partner Terragene. As the UK exclusive partner for the Infection Prevention giant, our portfolio includes:
- Protein Residue Testing
- Cleaning Indicators
- Chemical Indicators
- Biological Indicators
- Traceability Devices
Within the HTM01-01, there are several processes that are defined and must be carried as standard. Within Sychem’s vast portfolio, there are countless Infection Control consumables that can be used to test the process within Decontamination equipment, thus allowing their operation to continue.
Servicing
We offer a comprehensive range of equipment services for washing, disinfection, sterilisation, and decontamination equipment- designed to ensure that your system operates as efficiently as possible with minimal downtime.
At Sychem, we have a team of over 25 Engineers, based across the UK, and ensure that your equipment continues operating at it’s highest possible potential with weekly, quarterly, biannual and annual testing (testing requirements depend on the industry of operation).
Our services include the service and planned preventative maintenance (PPM) of equipment, validation & testing, refurbishment, emergency breakdown support and our unique cloud-based asset management portal, SYCHEMEYE.
We service all recognised brands of autoclaves, washer disinfectors, and other decontamination equipment. Our service contracts are fully customisable, and tailored to meet a facility’s needs. Our equipment services contracts can vary from periodic service & testing of equipment to fully comprehensive PPM contracts that incorporate breakdown cover, parts and training.

Contact a member of our expert team to find out more.
Can’t quite find what you’re looking for? Visit the Terragene website for more information.