ATP Monitoring Solutions
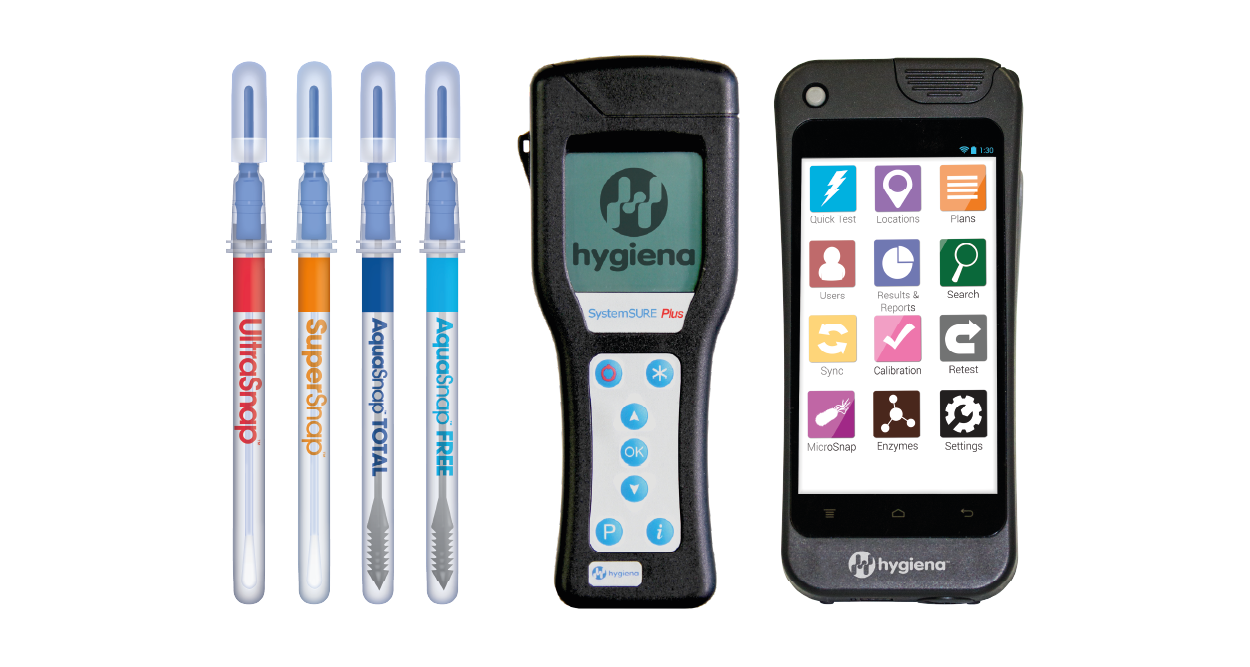
The SystemSURE Plus, the world’s best-selling ATP system, is known for its ease of use and reliability across a wide range of industries. The EnSURE Touch is a next-generation system with a 5-inch touchscreen, wireless syncing, cloud-based software, and the ability to conduct multiple tests, offering unmatched flexibility.
Both systems, paired with a range of sampling tests like UltraSnap and AquaSnap, are ideal for ensuring hygiene standards are met and maintained effectively.
Our solutions are trusted by industry leaders for critical processes in Infection Control & Life Science sectors:




Our ATP Hygiene Monitoring Range
Features & Benefits
Instant Results
ATP tests provide hygiene verification in as little as 10 to 15 seconds.
Quantitative Hygiene Assessment
Luminometers measure ATP levels for precise numerical values for cleanliness validation.
Industry-Compliant Quality
Manufactured under ISO 9001:2015 standards, with AOAC validation for reliable performance.